Bonds Form When Water Is Removed To Hold
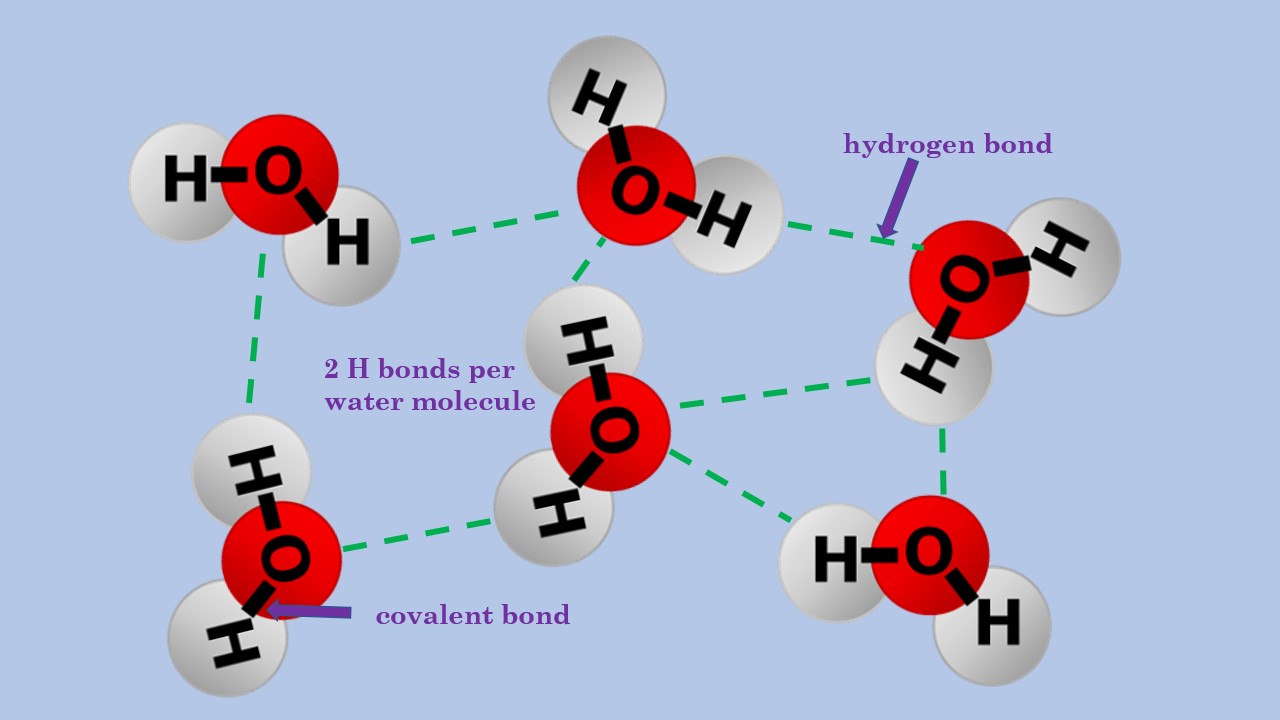
Bonds Form When Water Is Removed To Hold - Peptide bonds are covalent bonds that link amino acids. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. What is the effect of excess heat or temperature on an enzyme? Peptide bonds form when water is removed to hold amino acids together. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules.
Peptide bonds are covalent bonds that link amino acids. Peptide bonds form when water is removed to hold amino acids together. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. What is the effect of excess heat or temperature on an enzyme?
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. Peptide bonds form when water is removed to hold amino acids together. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. What is the effect of excess heat or temperature on an enzyme? A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds are covalent bonds that link amino acids.
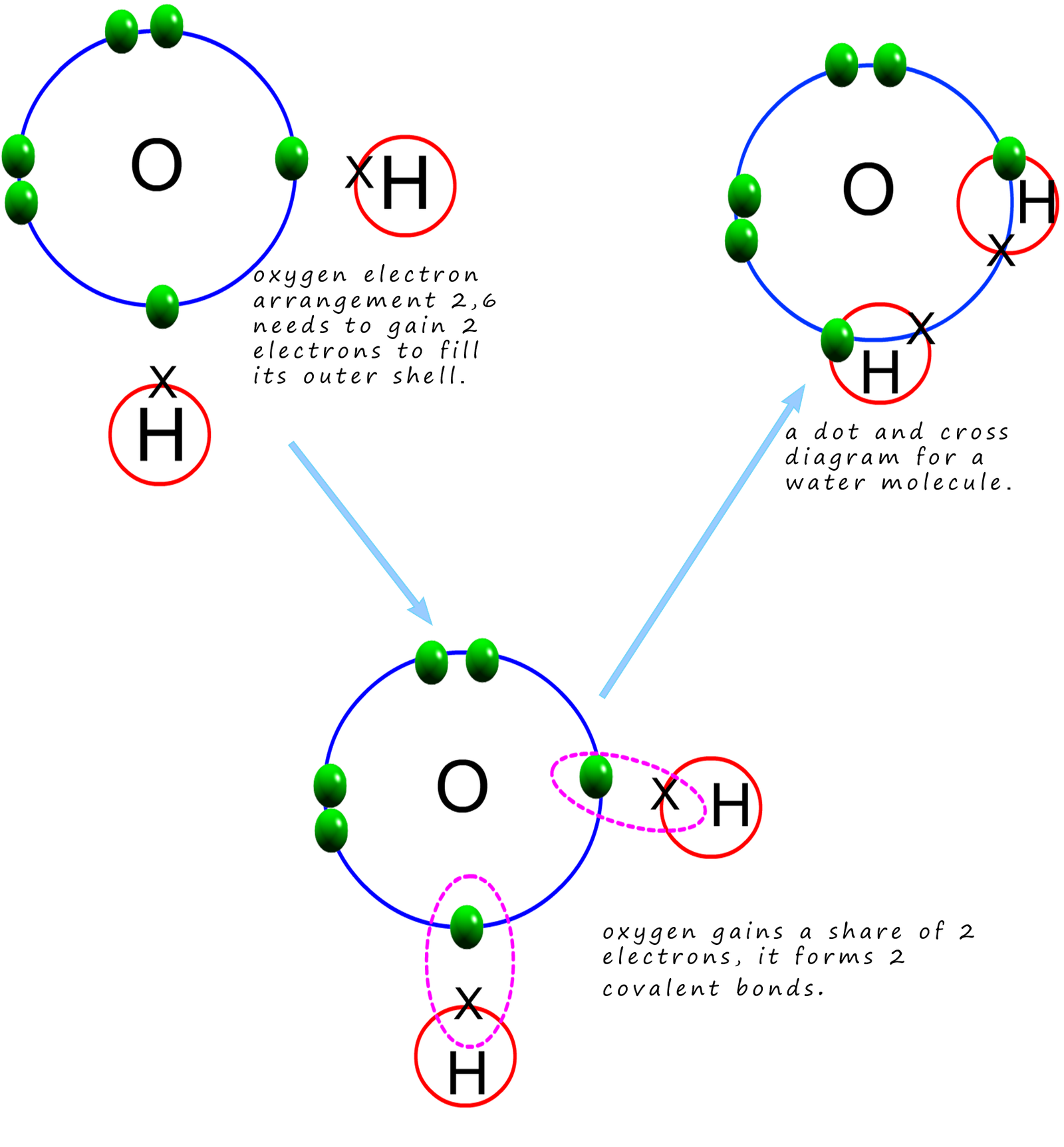
Covalent bonding
What is the effect of excess heat or temperature on an enzyme? In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. Peptide bonds form when.
[Solved] Label the bonds and components of this water molecule
A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Peptide bonds form when water is removed to hold amino acids together. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In water, each hydrogen nucleus is covalently.
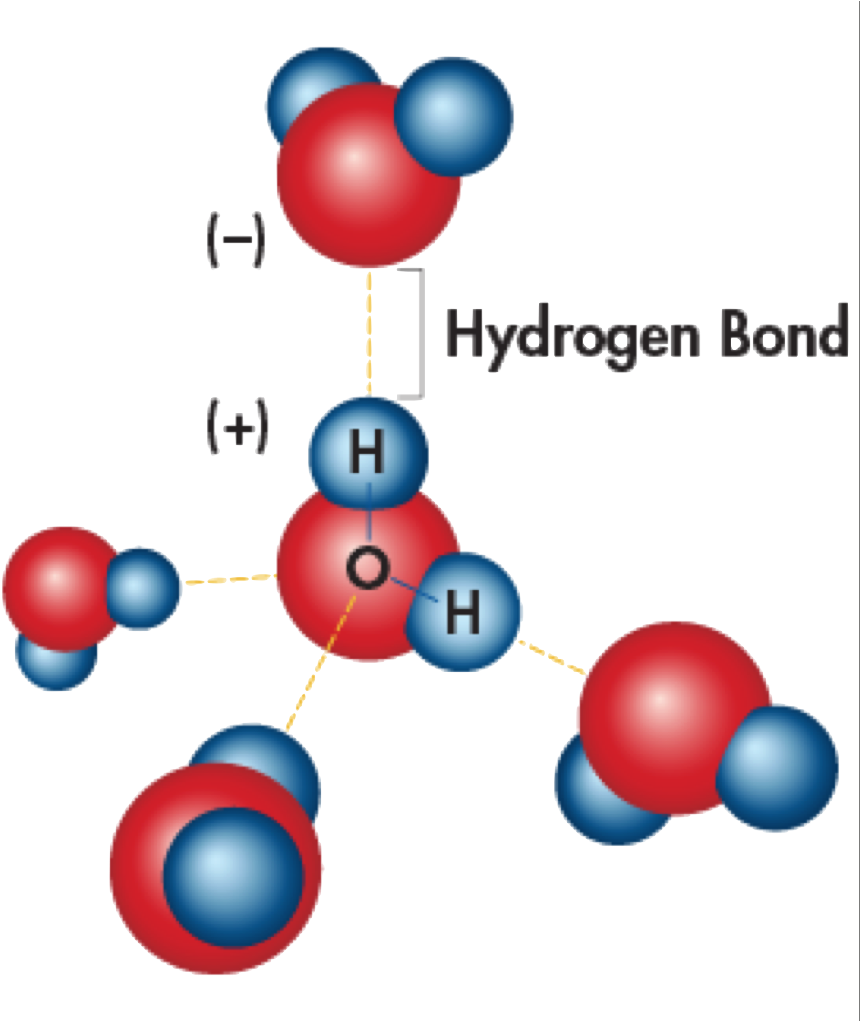
The diagram shows hydrogen bonds between water molecules. Label the
A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. Peptide bonds are covalent bonds that link amino acids. In the case of water, hydrogen bonds form between neighboring.
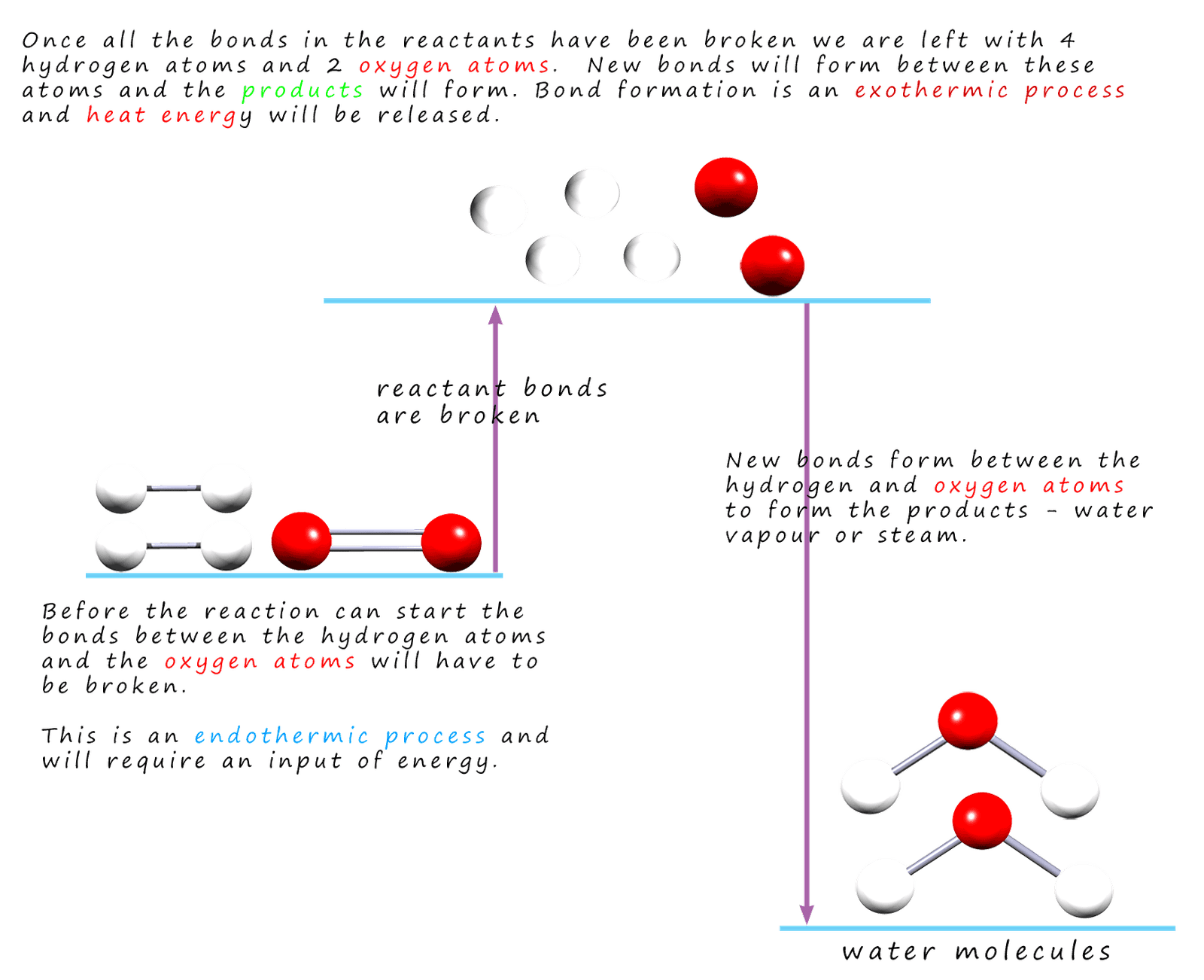
Bond energy
In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds form when water is removed to hold amino acids together. Peptide bonds are covalent bonds that link amino acids. What is the effect of excess heat or temperature on an enzyme? In the case of.
PPT Properties of Water PowerPoint Presentation, free download ID
The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. What is the effect of excess heat or temperature on an enzyme? Peptide bonds form when water is removed to hold amino acids together. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a.
Download Hydrogen Bonding Is The Effect Of Water Molecules Attracted
What is the effect of excess heat or temperature on an enzyme? In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds are covalent bonds that link.
Biomolecules flashcards 1 How do hydrogen bonds form between water
The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. What is the effect of excess heat or temperature on an enzyme? A drop of falling.
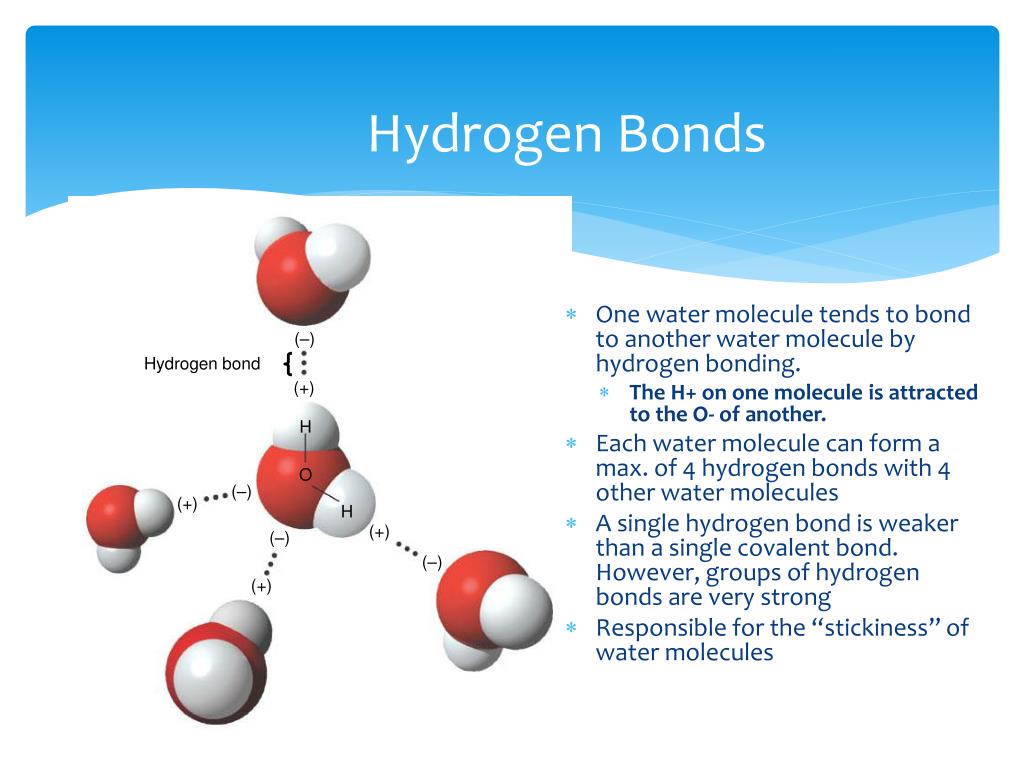
Hydrogen Bonding in water Dr. M. Chemistry Tutor
Peptide bonds form when water is removed to hold amino acids together. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds are covalent bonds that link amino acids. What is the effect of excess heat or temperature on an enzyme? The hydrogen bonds that.
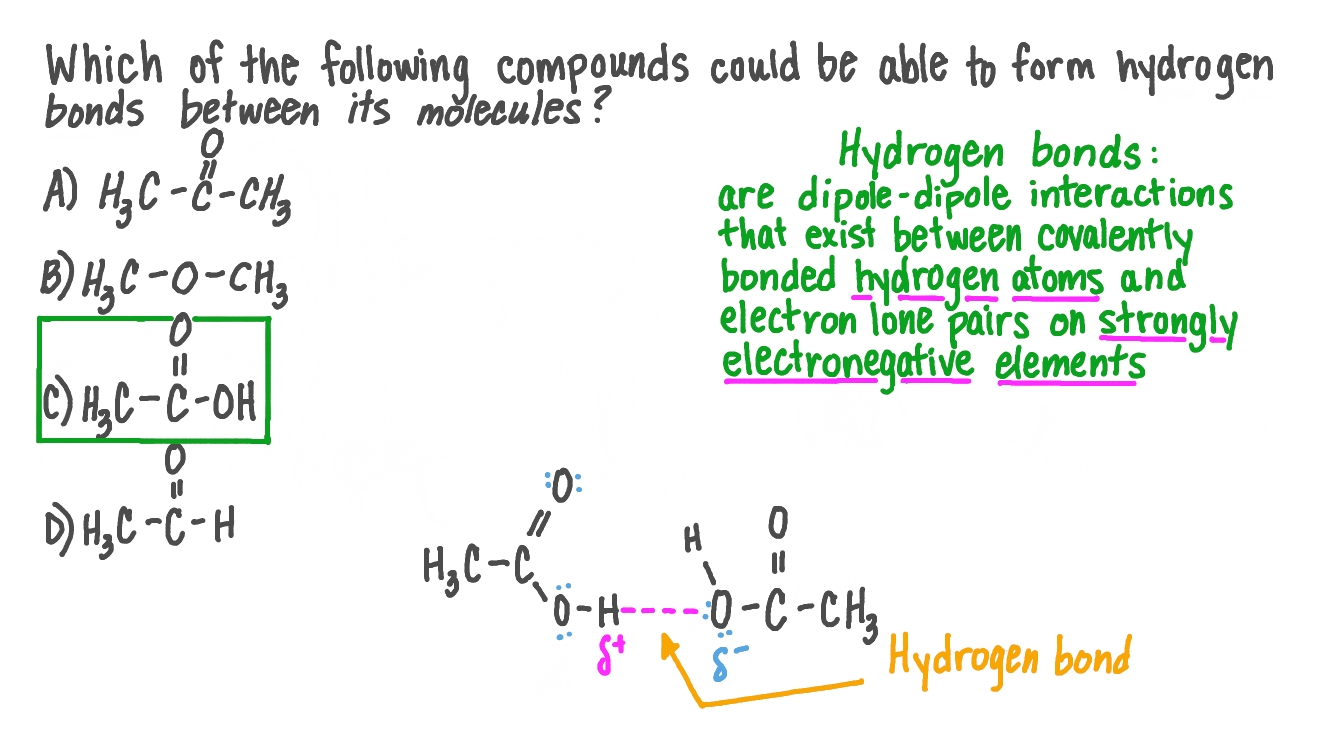
Question Video Identifying Which Compound Could Form Hydrogen Bonds
What is the effect of excess heat or temperature on an enzyme? Peptide bonds are covalent bonds that link amino acids. Peptide bonds form when water is removed to hold amino acids together. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. In the case of.
Primary and Secondary Bonds Owlcation
In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds are covalent bonds that link amino acids. What is the effect of excess heat or temperature on an enzyme? A drop of falling water is a group of water molecules held together by the hydrogen.
What Is The Effect Of Excess Heat Or Temperature On An Enzyme?
A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. Peptide bonds form when water is removed to hold amino acids together.
Peptide Bonds Are Covalent Bonds That Link Amino Acids.
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules.