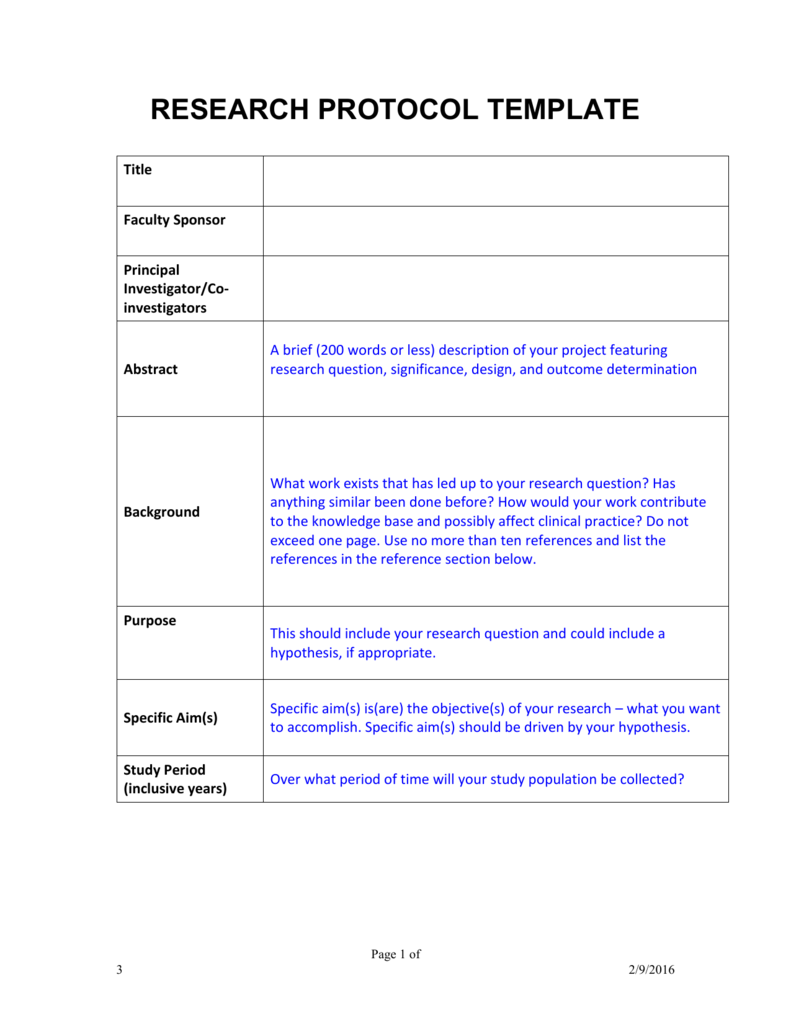
Clinical Research Protocol Template
Clinical Research Protocol Template - They follow the format of typical nih and industry multicenter protocols. Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. The natural history/observational protocol template, the. There are three templates to be used for observational research: The irb provides several protocol templates on this page. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research.
Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. They follow the format of typical nih and industry multicenter protocols. Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. The natural history/observational protocol template, the. Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. There are three templates to be used for observational research: The irb provides several protocol templates on this page. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data.
Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. There are three templates to be used for observational research: Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. The irb provides several protocol templates on this page. They follow the format of typical nih and industry multicenter protocols. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. The natural history/observational protocol template, the. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data.
Clinical Study Protocol (CSP) Template Clinical Study Templates
Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. The irb provides several protocol templates on this page. Protocol a written account of all the procedures to be.
research protocol template
The natural history/observational protocol template, the. Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. The irb provides several protocol templates on this page. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. They follow the.
Research Study Protocol Template
The natural history/observational protocol template, the. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. They follow the format of typical nih and industry multicenter protocols. The irb provides several protocol templates on this page. There are three templates to be used for observational research:
Clinical Research Protocol Template Edit Online & Download Example
Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. The irb provides several protocol templates on this page. Find free and adaptable templates and tools for.
Protocol Template Clinical Research Resource HUB
Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. They follow the format of typical nih and industry multicenter protocols. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. The natural history/observational protocol template, the. Research.
Protocol Template Treatment Use
Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. There are three templates to be used for observational research: Nih applicants can use a template with instructional and.
Clinical Protocol Template Master of Documents
Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. The irb provides several protocol templates on this page. The natural history/observational protocol template, the. They follow the format.
Study Protocol Template
Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. They follow the format of typical nih and industry multicenter protocols. Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. There are three templates to be.
Free Clinical Trial Templates Smartsheet
Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. The natural history/observational protocol template, the. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. Protocol a written account of all the procedures to be followed.
Phylotocol template. Based on the NIH clinical trial protocol, the
There are three templates to be used for observational research: They follow the format of typical nih and industry multicenter protocols. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. Protocol a written account of all the procedures to be followed in a trial, which describes all.
The Natural History/Observational Protocol Template, The.
There are three templates to be used for observational research: Research study protocol template (for clinical trials) instructions this protocol template is a tool to facilitate the development of a research. Protocol a written account of all the procedures to be followed in a trial, which describes all the administrative, documentation, analytical and. The irb provides several protocol templates on this page.
Nih Applicants Can Use A Template With Instructional And Sample Text To Help Write Clinical Protocols For The Following Types Of Research:.
Clinical trial protocol template this protocol template is designed to help research teams develop a clinical trial protocol that includes an. Find free and adaptable templates and tools for various aspects of clinical research, such as protocol development, informed consent, data. They follow the format of typical nih and industry multicenter protocols.