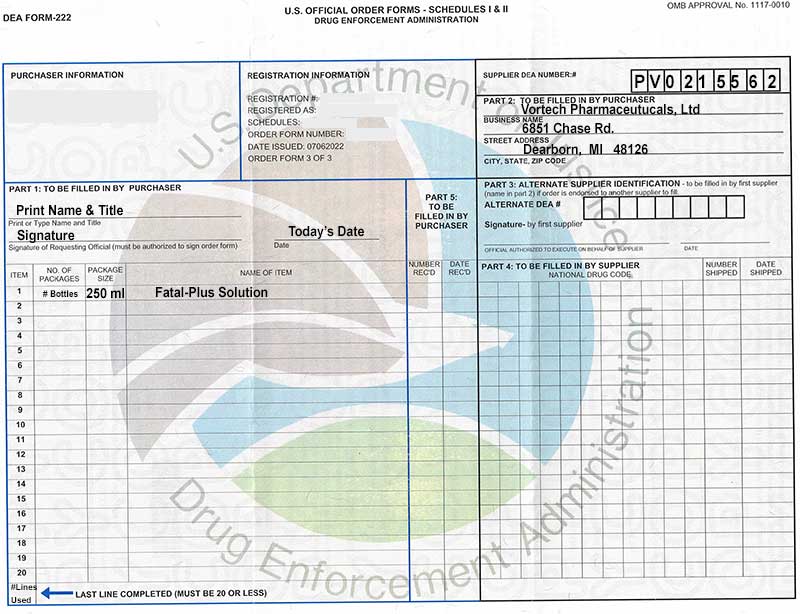
Dea 222 Form
Dea 222 Form - Modify dea registration to stop being a collector; Neither the controlled substances act nor its implementing regulations. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Under 21 cfr 1305.03, the completion of dea form 222 is required for each distribution of a schedule i or ii controlled substance. As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking.
Under 21 cfr 1305.03, the completion of dea form 222 is required for each distribution of a schedule i or ii controlled substance. As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); Neither the controlled substances act nor its implementing regulations. Modify dea registration to stop being a collector; The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking.
Modify dea registration to stop being a collector; The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Under 21 cfr 1305.03, the completion of dea form 222 is required for each distribution of a schedule i or ii controlled substance. Neither the controlled substances act nor its implementing regulations.
Fillable Online DEA 222 Form Single Sheet Instructions mmscms
Neither the controlled substances act nor its implementing regulations. As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution.
Filling out the new DEA 222 form for Pharmacy returns (as the Supplier
The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); Neither the controlled substances act nor its implementing regulations. Under 21 cfr 1305.03, the completion of dea form 222.
DEA form 222 A Guide to the Rules and Usage
As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. Neither the controlled substances act nor its implementing regulations. Under 21 cfr 1305.03, the completion of dea form 222 is required for each distribution of a schedule i.
How To Order Vortech Pharmaceuticals, Ltd.
The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Neither the controlled.
Printable New Dea 222 Form Printable Forms Free Online
Modify dea registration to stop being a collector; The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate.
Printable New Dea 222 Form Printable Forms Free Online
Neither the controlled substances act nor its implementing regulations. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the.
Dea 222 Form Example Complete with ease airSlate SignNow
As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance. Neither.
Medication Ordering
The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking. Modify dea registration to stop being a collector; The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule.
Dea Chapter 35 Application
As of october 30, 2021, the drug enforcement administration is implementing the mandatory use of a single sheet dea 222 order form and the triplicate dea order forms will no longer be. Neither the controlled substances act nor its implementing regulations. The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution.
MPS Example DEA 222 Form
The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The dea regulations provide that either a dea form 222 or its electronic equivalent is required.
Neither The Controlled Substances Act Nor Its Implementing Regulations.
Modify dea registration to stop being a collector; The drug enforcement administration (dea), office of diversion control, will accept requests from distributors that require a large volume of order forms (dea form 222) with the pin feed tracking. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The dea regulations provide that either a dea form 222 or its electronic equivalent is required for each distribution of a schedule i or ii controlled substance.
As Of October 30, 2021, The Drug Enforcement Administration Is Implementing The Mandatory Use Of A Single Sheet Dea 222 Order Form And The Triplicate Dea Order Forms Will No Longer Be.
Under 21 cfr 1305.03, the completion of dea form 222 is required for each distribution of a schedule i or ii controlled substance.