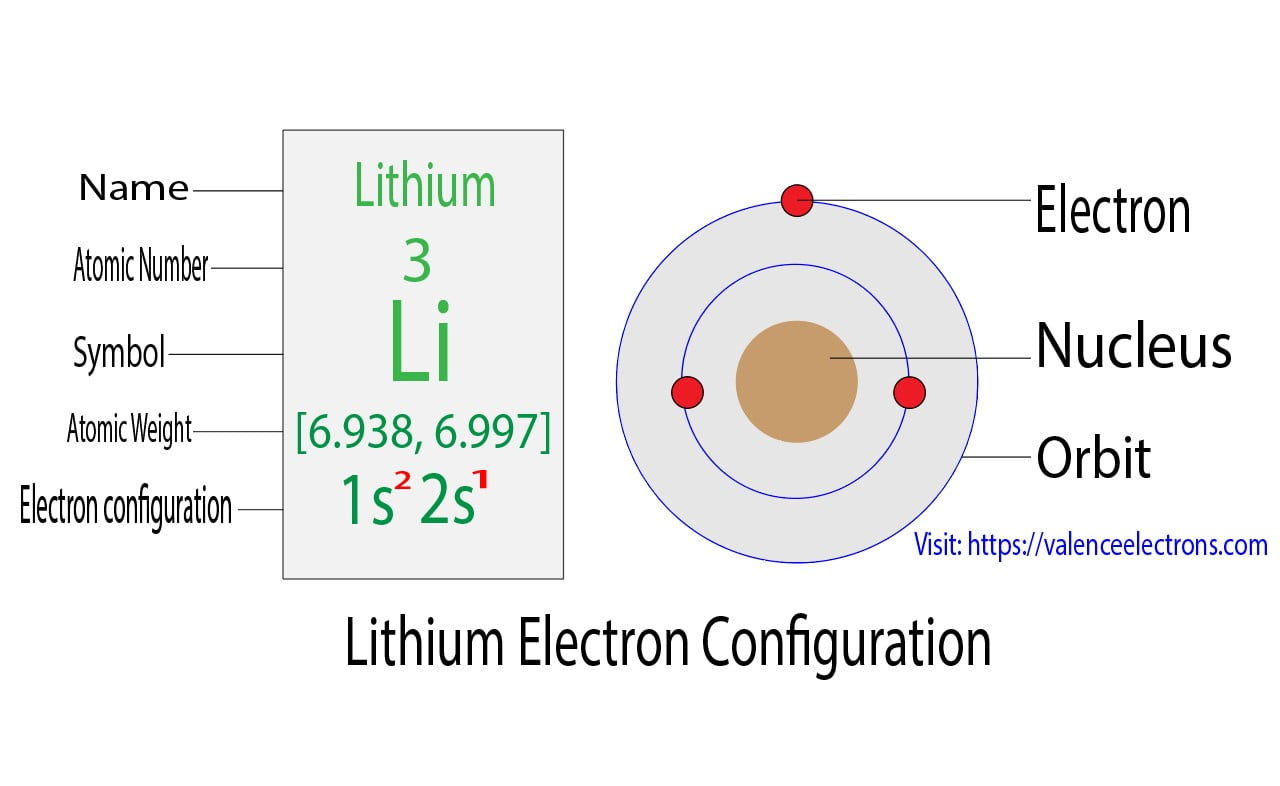
Lithium Electron Configuration Long Form
Lithium Electron Configuration Long Form - Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Access detailed info on all elements: View rotating bohr models for all 118. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Since 1s can hold only two electrons, the rest of. Atomic mass, electron configurations, charges, and more. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom.
The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Access detailed info on all elements: View rotating bohr models for all 118. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Since 1s can hold only two electrons, the rest of. Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Atomic mass, electron configurations, charges, and more.
View rotating bohr models for all 118. Since 1s can hold only two electrons, the rest of. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Access detailed info on all elements: Atomic mass, electron configurations, charges, and more.
Electron Configuration Of Lithium
Atomic mass, electron configurations, charges, and more. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Access detailed info on all elements: Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. To write the.
Lithium Atom Diagram / Atomic Structure Chemistry 10 A lithium atom
Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Since 1s can hold only two electrons, the rest of. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Atomic mass, electron configurations, charges, and.
Lithium electron configuration Stock Image C029/5021 Science
Since 1s can hold only two electrons, the rest of. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Atomic mass, electron configurations, charges, and more. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Now, the lithium cation, li+,.
Lithium Element With Reactions, Properties, Uses, & Price Periodic Table
Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Access detailed info on all elements: To write the electron configuration for lithium, the first two electrons enter the 1s orbital. View rotating bohr models for all 118. Atomic mass, electron configurations, charges, and more.
electronarrangementforlithiumatom TechnoCrazed
Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Atomic mass, electron configurations, charges, and more. Since 1s can hold only two electrons, the rest of. Access detailed info on all elements: View rotating bohr models for all 118.
Electron configurations
The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Atomic.
【5 Steps】Electron Configuration of Lithium(Li) in Just 5 Steps
Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Since 1s can hold only two electrons, the rest of. To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Atomic mass, electron configurations, charges, and more. Access detailed info on all elements:
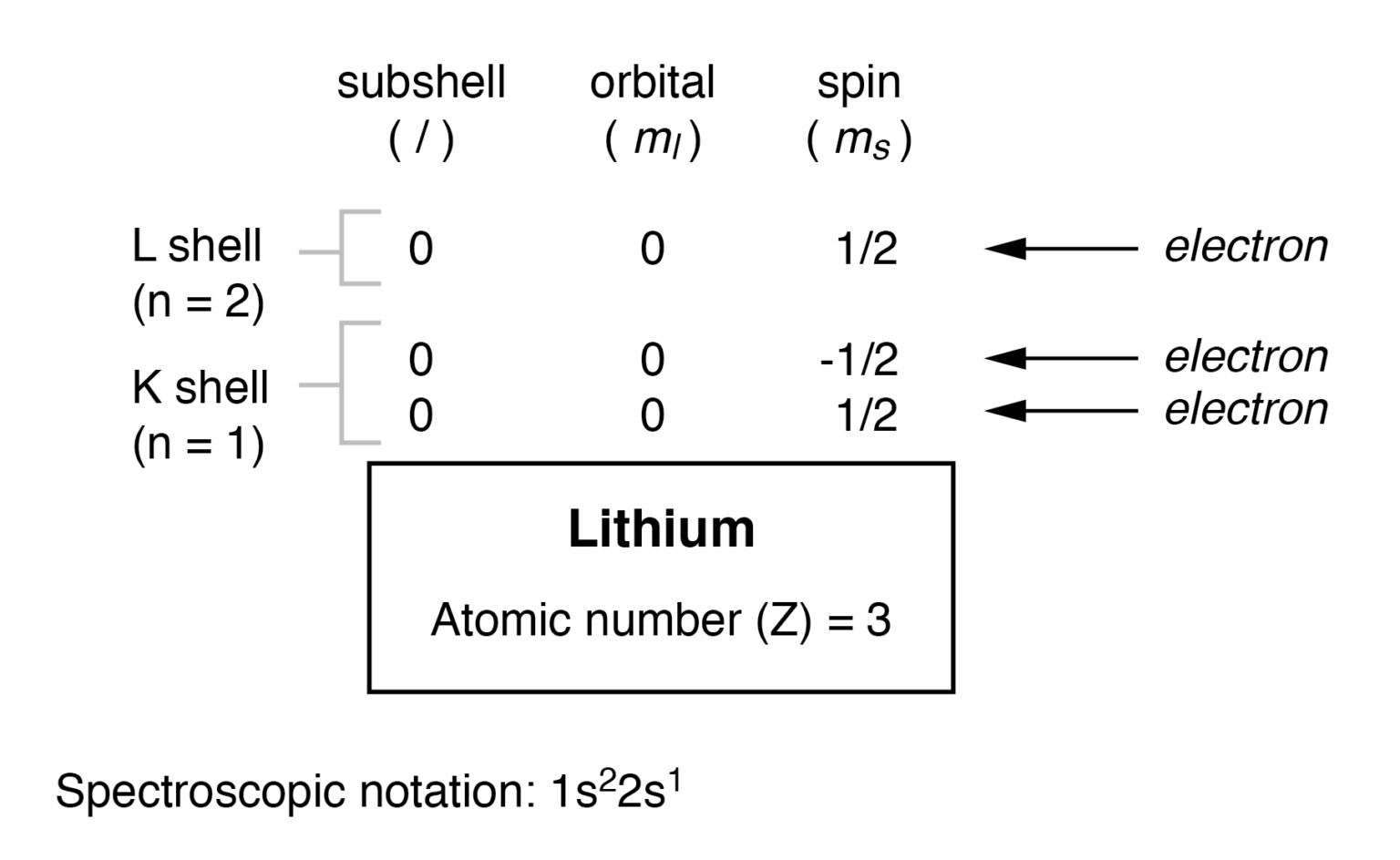
How to Write the Electron Configuration for Lithium?
View rotating bohr models for all 118. Access detailed info on all elements: Now, the lithium cation, li+, is formed when lithium loses the electron located on its outermost shell → its valence electron. Atomic mass, electron configurations, charges, and more. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the.
How Do We Can Find A Electron Configuration For Lithium
To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Since 1s can hold only two electrons, the rest of. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. Atomic mass, electron configurations, charges, and more. Now, the lithium cation, li+,.
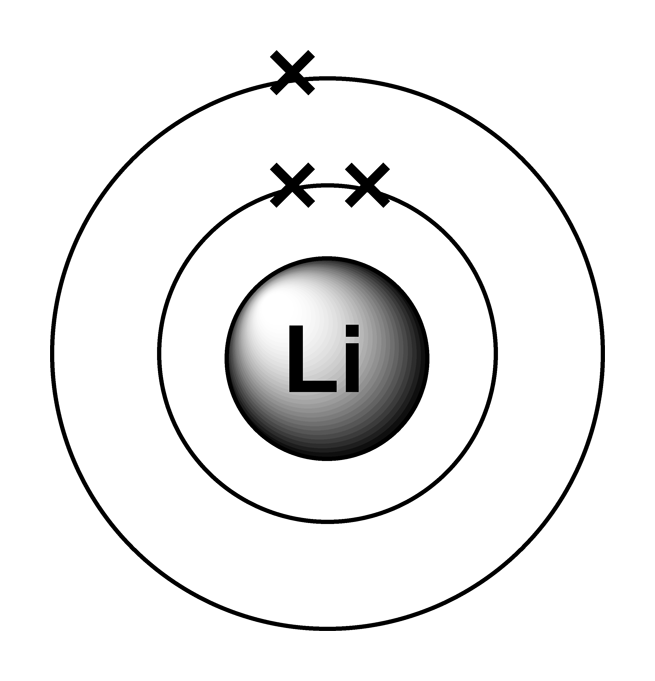
14+ Lithium Lewis Dot Structure Robhosking Diagram
Atomic mass, electron configurations, charges, and more. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. View rotating bohr models for all 118. Since 1s can hold only two electrons, the rest of. To write the electron configuration for lithium, the first two electrons enter the.
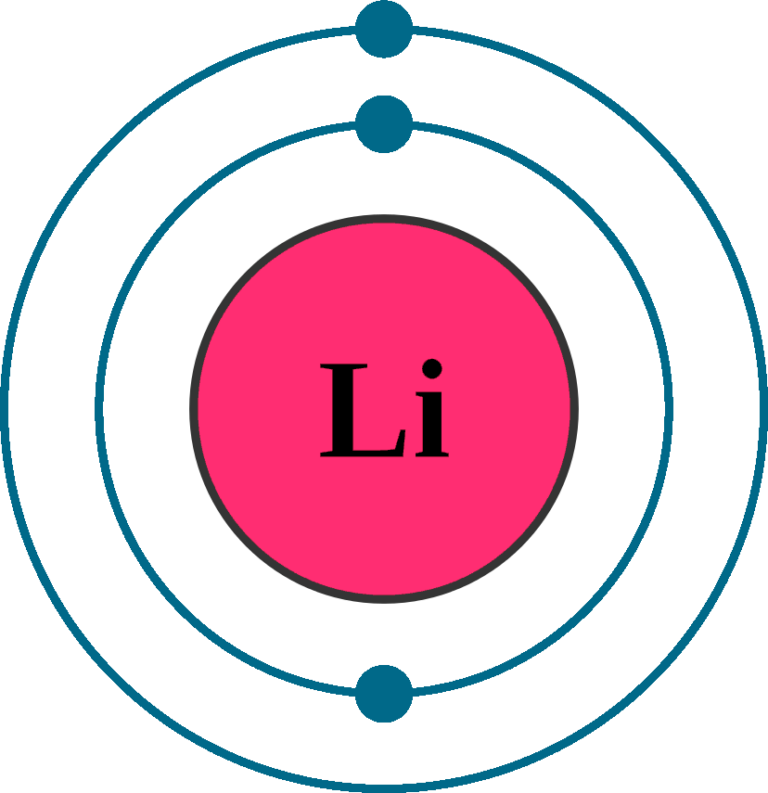
Now, The Lithium Cation, Li+, Is Formed When Lithium Loses The Electron Located On Its Outermost Shell → Its Valence Electron.
To write the electron configuration for lithium, the first two electrons enter the 1s orbital. Atomic mass, electron configurations, charges, and more. View rotating bohr models for all 118. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom.
Since 1S Can Hold Only Two Electrons, The Rest Of.
Access detailed info on all elements:





/lithiumatom-56a12c335f9b58b7d0bcc103.jpg)