What Is The Electron Configuration Of Br
What Is The Electron Configuration Of Br - Atomic mass, electron configurations, charges, and more. These blocks are named for the characteristic spectra. Bromine belongs to group 17 which is known as halogens. The magnetic form of a substance can be determined by examining its electron configuration: Access detailed info on all elements: If it shows unpaired electrons, then the substance. Elements are organised into blocks by the orbital type in which the outer electrons are found. View rotating bohr models for all 118.
Elements are organised into blocks by the orbital type in which the outer electrons are found. Access detailed info on all elements: If it shows unpaired electrons, then the substance. These blocks are named for the characteristic spectra. The magnetic form of a substance can be determined by examining its electron configuration: Atomic mass, electron configurations, charges, and more. View rotating bohr models for all 118. Bromine belongs to group 17 which is known as halogens.
Bromine belongs to group 17 which is known as halogens. View rotating bohr models for all 118. Atomic mass, electron configurations, charges, and more. Access detailed info on all elements: The magnetic form of a substance can be determined by examining its electron configuration: Elements are organised into blocks by the orbital type in which the outer electrons are found. If it shows unpaired electrons, then the substance. These blocks are named for the characteristic spectra.
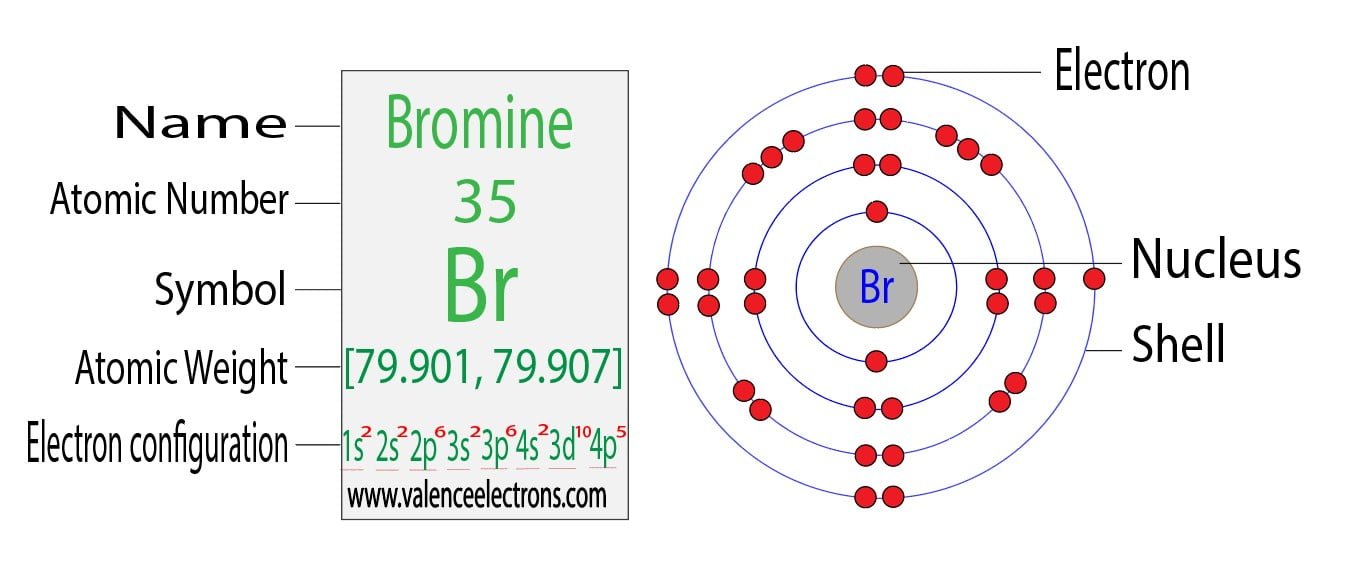
Bromine Electron Configuration (Br) with Orbital Diagram
Elements are organised into blocks by the orbital type in which the outer electrons are found. If it shows unpaired electrons, then the substance. The magnetic form of a substance can be determined by examining its electron configuration: View rotating bohr models for all 118. Access detailed info on all elements:
Br Electron Configuration (Bromide Ion) YouTube
These blocks are named for the characteristic spectra. View rotating bohr models for all 118. Elements are organised into blocks by the orbital type in which the outer electrons are found. Bromine belongs to group 17 which is known as halogens. Access detailed info on all elements:
Electron Configuration Magnesium
If it shows unpaired electrons, then the substance. View rotating bohr models for all 118. The magnetic form of a substance can be determined by examining its electron configuration: Atomic mass, electron configurations, charges, and more. Bromine belongs to group 17 which is known as halogens.
Electron Configuration For Bromine
These blocks are named for the characteristic spectra. Access detailed info on all elements: Elements are organised into blocks by the orbital type in which the outer electrons are found. Bromine belongs to group 17 which is known as halogens. If it shows unpaired electrons, then the substance.
Cr2+ Ground State Electron Configuration Electron Configurations
If it shows unpaired electrons, then the substance. Access detailed info on all elements: The magnetic form of a substance can be determined by examining its electron configuration: View rotating bohr models for all 118. Atomic mass, electron configurations, charges, and more.
Electronic Configurations Intro Chemistry LibreTexts
View rotating bohr models for all 118. Atomic mass, electron configurations, charges, and more. Elements are organised into blocks by the orbital type in which the outer electrons are found. Bromine belongs to group 17 which is known as halogens. The magnetic form of a substance can be determined by examining its electron configuration:
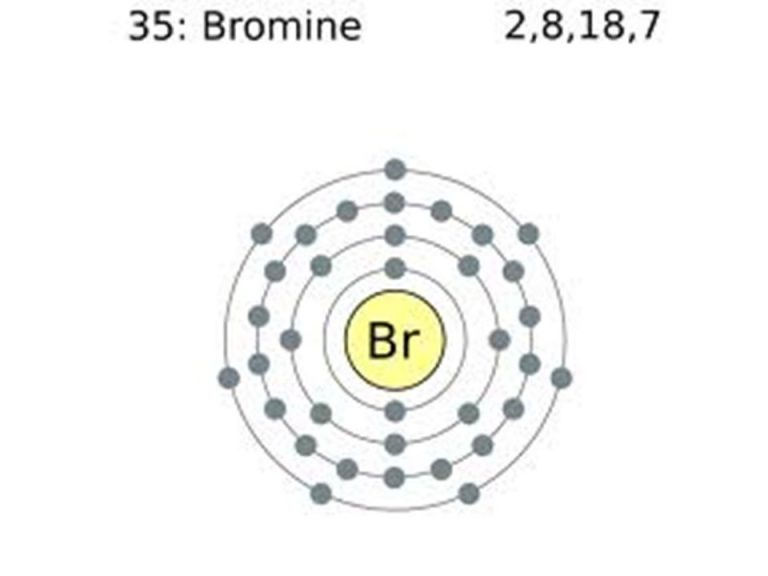
Atom Diagrams Electron Configurations of the Elements
These blocks are named for the characteristic spectra. Access detailed info on all elements: If it shows unpaired electrons, then the substance. Bromine belongs to group 17 which is known as halogens. The magnetic form of a substance can be determined by examining its electron configuration:
Bromine Electron Configuration (Br) with Orbital Diagram
If it shows unpaired electrons, then the substance. Elements are organised into blocks by the orbital type in which the outer electrons are found. Access detailed info on all elements: The magnetic form of a substance can be determined by examining its electron configuration: Bromine belongs to group 17 which is known as halogens.
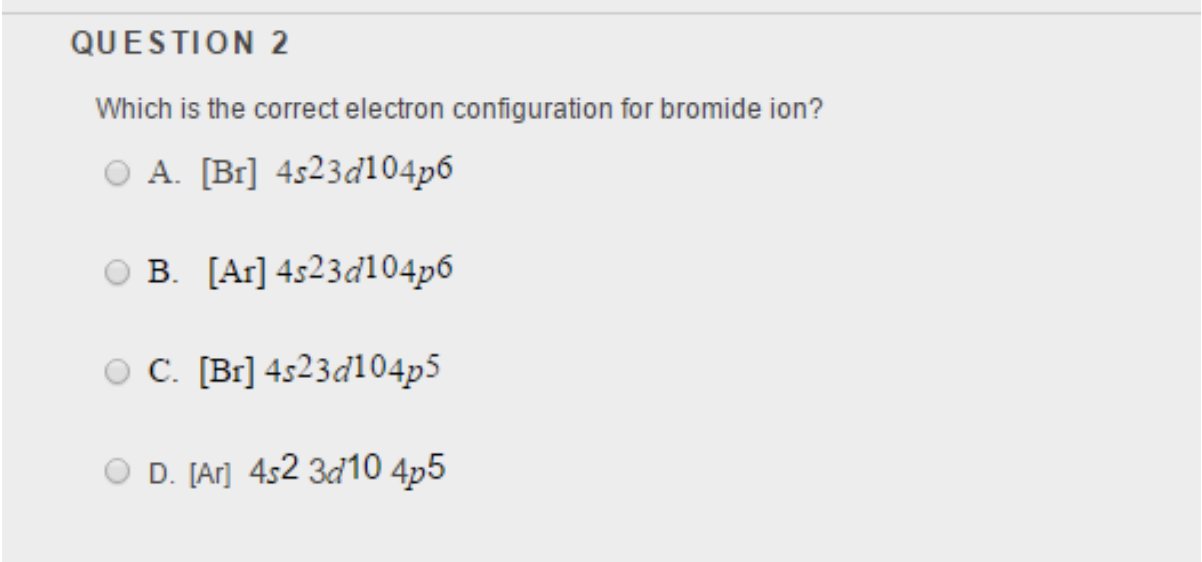
Solved QUESTION 2 Which is the correct electron
If it shows unpaired electrons, then the substance. Access detailed info on all elements: Bromine belongs to group 17 which is known as halogens. The magnetic form of a substance can be determined by examining its electron configuration: View rotating bohr models for all 118.
Electron Configuration For Bromine
Bromine belongs to group 17 which is known as halogens. The magnetic form of a substance can be determined by examining its electron configuration: These blocks are named for the characteristic spectra. Access detailed info on all elements: Atomic mass, electron configurations, charges, and more.
View Rotating Bohr Models For All 118.
Atomic mass, electron configurations, charges, and more. Access detailed info on all elements: The magnetic form of a substance can be determined by examining its electron configuration: Elements are organised into blocks by the orbital type in which the outer electrons are found.
If It Shows Unpaired Electrons, Then The Substance.
These blocks are named for the characteristic spectra. Bromine belongs to group 17 which is known as halogens.





:max_bytes(150000):strip_icc()/Bromine-58b601f93df78cdcd83d2817.jpg)