What Is The Value Of The Bond Angles In Sicl4
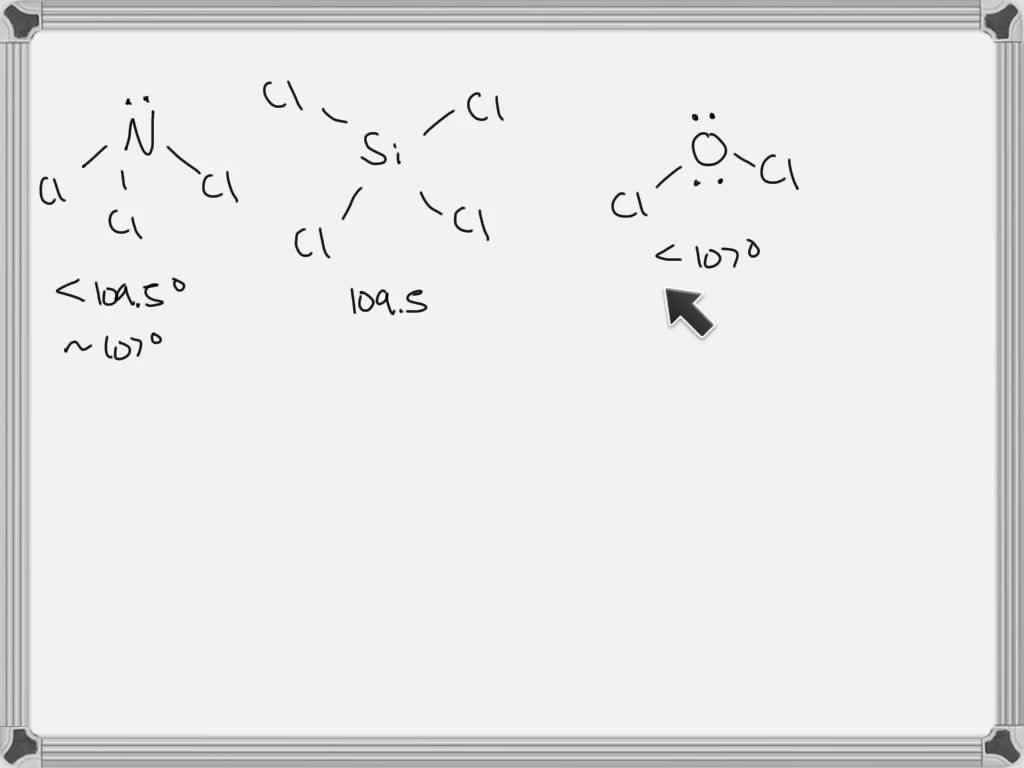
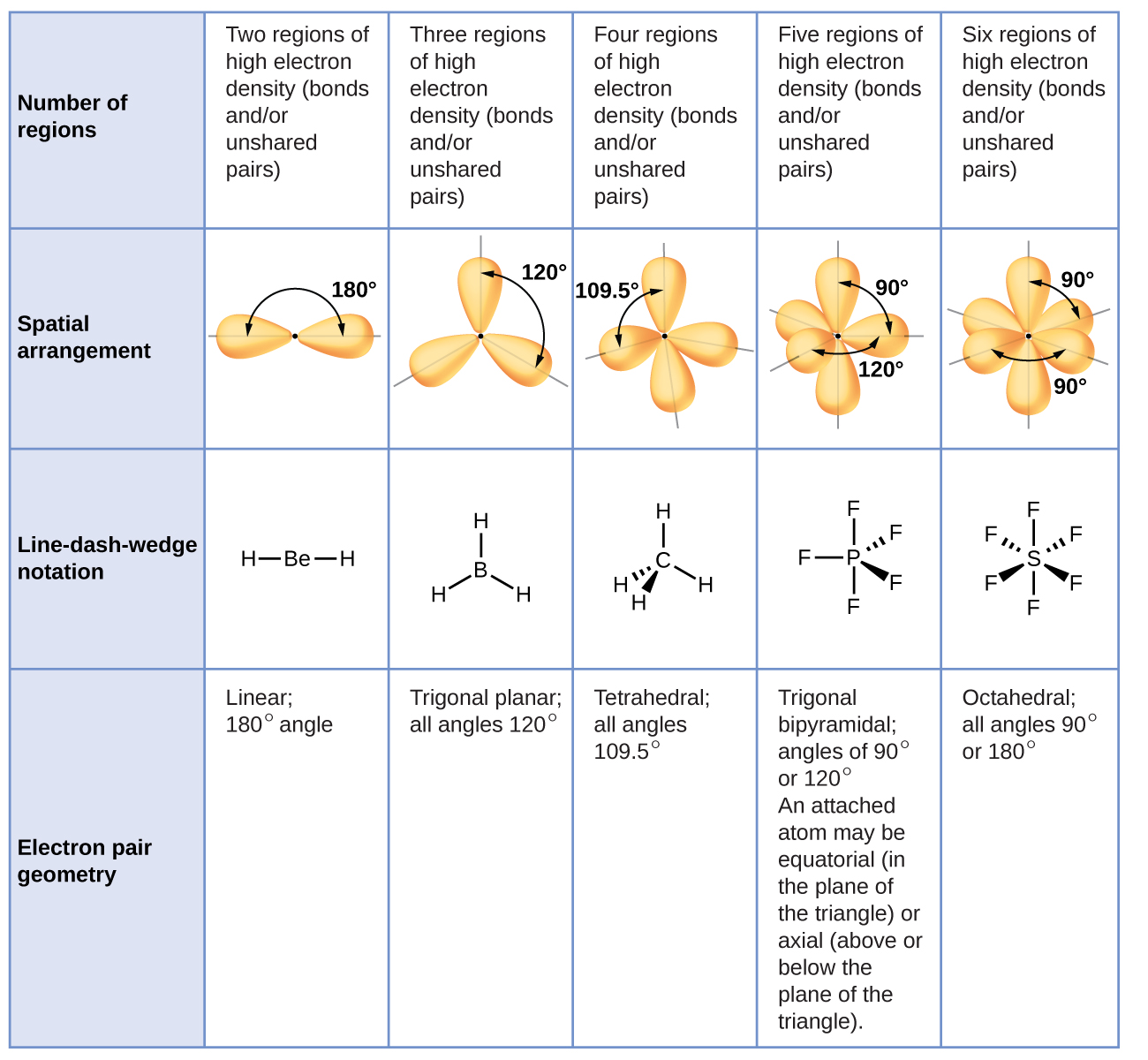
What Is The Value Of The Bond Angles In Sicl4 - Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. What is the value of the bond angles in sicl_(4) ? The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: What is the value of the bond angles in sicl_(4) ? Sicl4 molecular and electron geometry.
What is the value of the bond angles in sicl_(4) ? Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. Sicl4 molecular and electron geometry. What is the value of the bond angles in sicl_(4) ? The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below:
The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: Sicl4 molecular and electron geometry. The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. What is the value of the bond angles in sicl_(4) ? Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. What is the value of the bond angles in sicl_(4) ?
Scl4 Lewis Structure Molecular Geometry
What is the value of the bond angles in sicl_(4) ? The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. The structure of s i c l.
Solved d. Use data about solid SiCl4 to estimate a value for
Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. Sicl4 molecular and electron geometry. The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a.
SOLVED The correct order of increasing ClXCl bond angles is I. NCl3
Sicl4 molecular and electron geometry. The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: What is the value of the bond angles in sicl_(4) ? The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. What is the.
29+ how to calculate bond angles FerdavsAlaa
The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: Sicl4 molecular and electron geometry. The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles.
predict the approximate bond angle in the following molecule.
The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: Sicl4 molecular and electron geometry. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a.
SOLVED Chemistry 130 Experiment 12 Chemical Bonding Formulas SiCl4
Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. What is.
Diagram of Bond Angles Quizlet
Sicl4 molecular and electron geometry. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. What is the value of the bond angles in sicl_(4) ? The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: What is the value of the bond angles in sicl_(4) ?
SOLVEDFor each of the following species, write the Lewis structure
Sicl4 molecular and electron geometry. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. What is the value of the bond angles in sicl_(4) ? The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: The bond angle of sicl4 is 109.5° as the shape of.
SOLVED The molecules SiCl4 and PCl3 have the same electrondomain
The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below: The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. Sicl4 molecular.
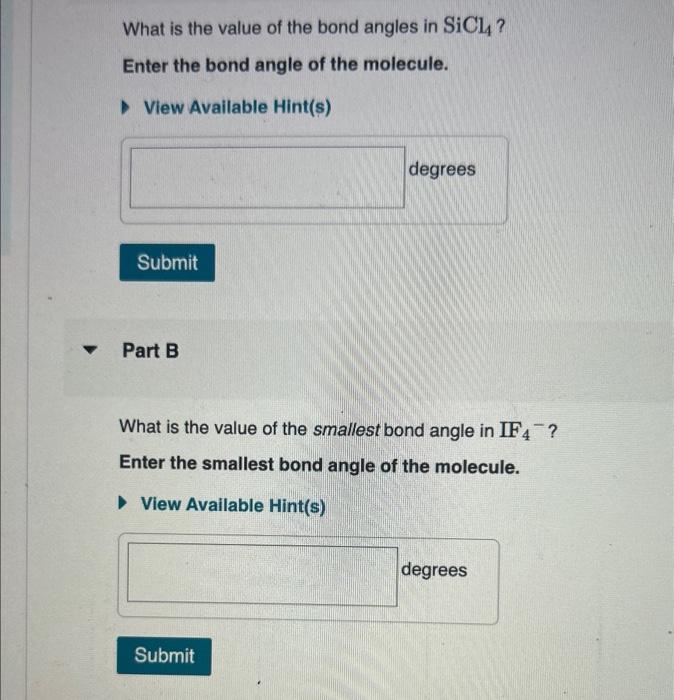
Solved What is the value of the bond angles in SiCl4 ? Enter
What is the value of the bond angles in sicl_(4) ? Sicl4 molecular and electron geometry. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. What is the value of the bond angles in sicl_(4) ? The structure of s i c l x 4 \ce{sicl4} sicl x 4 is given below:
The Structure Of S I C L X 4 \Ce{Sicl4} Sicl X 4 Is Given Below:
What is the value of the bond angles in sicl_(4) ? Sicl4 molecular and electron geometry. Part a) for sicl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.5 degrees. The bond angle of sicl4 is 109.5° as the shape of its tetrahedral in nature and as per the vsepr theory, a regular tetrahedral molecule.